Products

Utilizing our resin blending and co-extrusion multi-layer film processing
technology, this is an easily recyclable film made of a single polyethylene material.
By using a single material, recyclability is improved, while the material also has heat resistance,
easy peelability, pinhole resistance and thermo-formability.
Plant
- Sumitomo Bakelite (Nantong) Co., Ltd.
Laboratory
- Films & Sheets Research Laboratory
Features
As made of a single polyethylene material, there is no need to separate it into an individual material after use, making it suitable for recycling.
By combining polyethylene-based materials with different properties, it has properties equal to or greater than those of conventional composite material products.
CEL-9530A has been certified as compatible with readily available sorting & recycling technologies
commonly used at industrial scale in the recycling sector in Europe through the assessment under the
RecyClass Recyclability Methodology.
It has also been confirmed that CEL-9530A can be sorted into the polyethylene film-fraction (Assessment
as an intermediate product that constitutes the final package).
【Certification code: ry655-sbc-ts】
Changes in design, printing, used materials, and the effect of product content or residue might change the outcome of the assessment of a final packaging using SUMILITE™ CEL-9530A. The Letter of Compatibility class result offers scoring on recyclability in Europe, which does not consider the specific collection systems in the different countries.
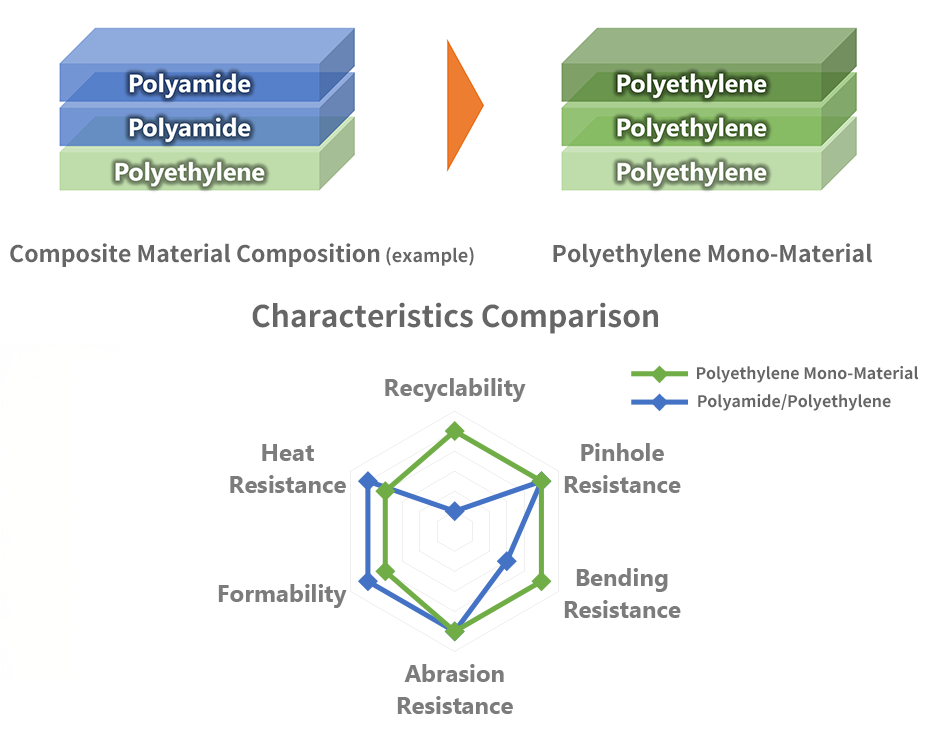
Note:
- This graph shows a relative comparison of the properties of each film, and does not indicate absolute property values.
Applications
Medical Device Packaging
Medical Device Packaging Materials

Specifications
| Items | Test method | Unit | Direction | CEL-9530A |
| Thickness | - | µm | - | 150 |
| Tensile Strength | JIS K 7127 | N/mm2 | MD | 49 |
| TD | 43 | |||
| Elongation | JIS K 7127 | % | MD | 776 |
| TD | 762 | |||
| Tear strength | JIS K 7128 | N/cm | MD | 1198 |
| TD | 1170 | |||
| Light transmittance | JIS K 7361 | % | - | 89 |
| Haze | JIS K 7136 | % | - | 15 |
Remark
- The values mentioned above are typical, not guaranteed.
- 2026/04/01ProductsLaunch of Official Production of the High-speed Sealing Cover Tape SUMILITE™ CSL-Z7304
- 2026/02/20Products Obtained Recyclability Certification for Medical Device Packaging Film
- 2025/04/10ProductsDevelopment of Medical Device Packaging Mono-material Film that is Easy to Recycle
- 2023/10/13ProductsAcquisition of Film Business of Asahi Kasei Pax Corp.

 Inquiry
Inquiry