Sumitomo Bakelite Co., Ltd.
SB-KAWASUMI LABORATORIES, INC.
Akita Sumitomo Bakelite Co., Ltd.
April 26, 2026
Sumitomo Bakelite Co., Ltd. (Headquarters: Shinagawa-ku, Tokyo; President and Representative Director: Shinichi Kajiya) announced its decision to expand production facilities for microcatheters at Akita Sumitomo Bakelite Co., Ltd. in response to the continued global growth in demand for steerable microcatheters used in endovascular treatment.
This product is a new type of microcatheter that allows physicians to arbitrarily control the direction of its tip through manual manipulation, thereby contributing to a reduction in procedure time. Since its launch in 2016, it has accumulated a strong track record in clinical settings both in Japan and abroad, aligned with advances in the sophistication and minimally invasive nature of endovascular treatments.
Changes in the Environment Surrounding Endovascular Therapy and the Role of Microcatheters
As the population ages, the risk of vascular diseases such as arteriosclerosis and vascular stenosis, as well as diseases caused by lifestyle-related conditions, is increasing year by year. In addition, the demand for minimally invasive treatments, including endovascular therapy, is growing as treatment options applicable to elderly patients and those with comorbidities.
Minimally invasive treatments are becoming more widespread as they impose less physical burden compared to surgical procedures and are expected to shorten hospital stays and enhance patients' quality of life.
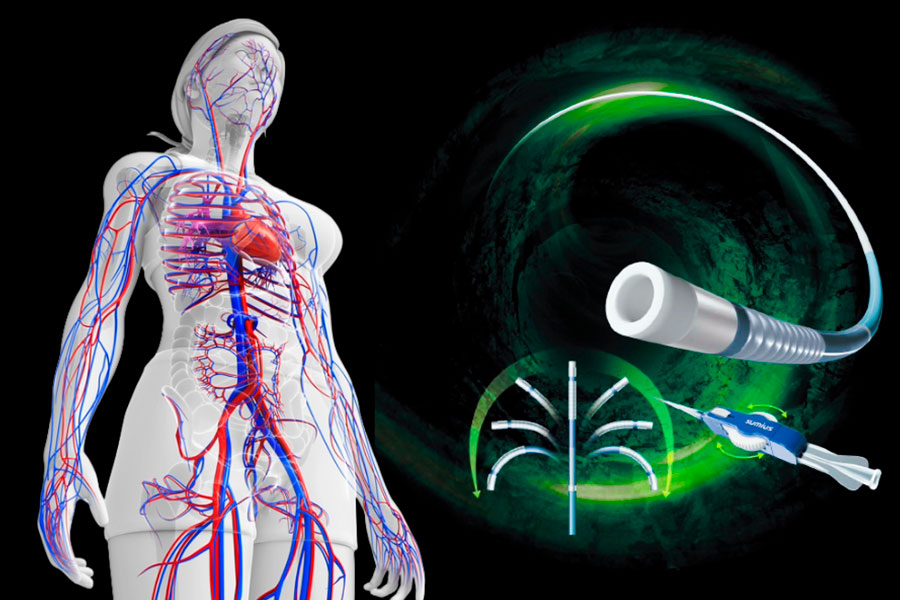
One of the essential devices for such treatments is the microcatheter, which is used across a wide range of areas including cerebrovascular, abdominal, and peripheral vasculature. Along with advances in ancillary devices, the demand for higher performance microcatheters is increasing as the need to address more complex and advanced cases grows.
Features and Clinical Value of the Steerable Microcatheter
The Steerable Microcatheter features a design that allows arbitrary control of the tip direction through manual operation. This articulating tip mechanism enables selective and stable navigation even in highly tortuous vessels and complex vascular branches, which are difficult to approach with conventional microcatheters.
Our steerable microcatheter is selected for a variety of conditions, including embolization therapy for tumors such as hepatocellular carcinoma in the abdominal region, coil embolization of cerebral aneurysms in the neurovascular region, and treatment of ischemic diseases in the peripheral vascular region.
The articulating tip feature of this product enhances accessibility to target vessels, facilitating easier vessel selection and reducing the operational burden on physicians, while contributing to shorter procedure times. This, in turn, is expected to reduce fluoroscopy time and the number of contrast injections, leading to decreased radiation exposure not only for patients but also for healthcare professionals.
Device Name: SwiftNINJA™ Steerable Microcatheter
Since its launch, the indications for the product have expanded in response to advances in medical technology and changing clinical needs. Our company has continuously enhanced the product line by expanding size variations and making ongoing improvements both domestically and internationally. In Japan, in 2022, we obtained regulatory approval for indications targeting intracranial endovascular therapy and achieved an expansion of insurance coverage, thereby aligning our product development closely with clinical demands.
Thanks to these features, the Steerable Microcatheter has become established in medical settings as a reliable tool supporting safer and more accurate endovascular treatments. Nearly ten years after the initial launch, demand for the product continues to grow.
Overview of Production Expansion
The production expansion will be implemented at Akita Sumitomo Bakelite Co., Ltd. and is intended to strengthen the stable supply of steerable microcatheters to medical institutions worldwide in response to steadily growing demand. Preparation for equipment installation will begin in April 2026, with full-scale operation scheduled to commence in February 2027.
In Japan, the product is marketed by SB-KAWASUMI LABORATORIES, INC., a member of our group. Overseas, the product is distributed through a partner company and is currently available in approximately 40 countries worldwide, primarily in the United States and China.
Through this enhancement of the production system, the company expects production capacity in fiscal year 2027 to reach approximately 200% of fiscal year 2025 levels, supporting further expansion in global markets.
Akita Sumitomo Bakelite Co., Ltd.

President: Goichiro Kuwaki
Head Office: 27-4 Nakajima-shita, Aisome-machi, Tsuchizaki-Minato, Akita City,
Akita 011-8510, Japan
Year Established: 1970
Number of Employees: 179
Business Overview: Manufacture of medical devices, bio-related products, phenolic resins, and other advanced materials.
SB-KAWASUMI LABORATORIES, INC.

Representative Director, President & COO: Akiko Okubo
Head Office: 3-25-4 Tonomachi, Kawasaki-ku, Kawasaki City, Kanagawa 210-8602, Japan
Year Established: 2021 Number of Employees: 1,811
Business Overview: Development, manufacturing, and sales of medical devices and pharmaceutical products.
Looking Ahead
Our Group remains committed to addressing evolving clinical needs in minimally invasive therapy through the development and reliable supply of high-quality medical devices. This expansion of production capacity represents an important initiative to fulfill its responsibility as a medical device manufacturer, ensuring stable supply, consistent quality, and continued improvement in support of physicians and patients worldwide.
Related Information
![]() Expansion of Production Capacity for Steerable Microcatheters(PDF 379KB)
Expansion of Production Capacity for Steerable Microcatheters(PDF 379KB)
Contact Information
Endovascular Products Sales Department, Domestic Sales Division, SB-KAWASUMI LABORTORIES, Inc.
Tel: 81-3-5462-4824